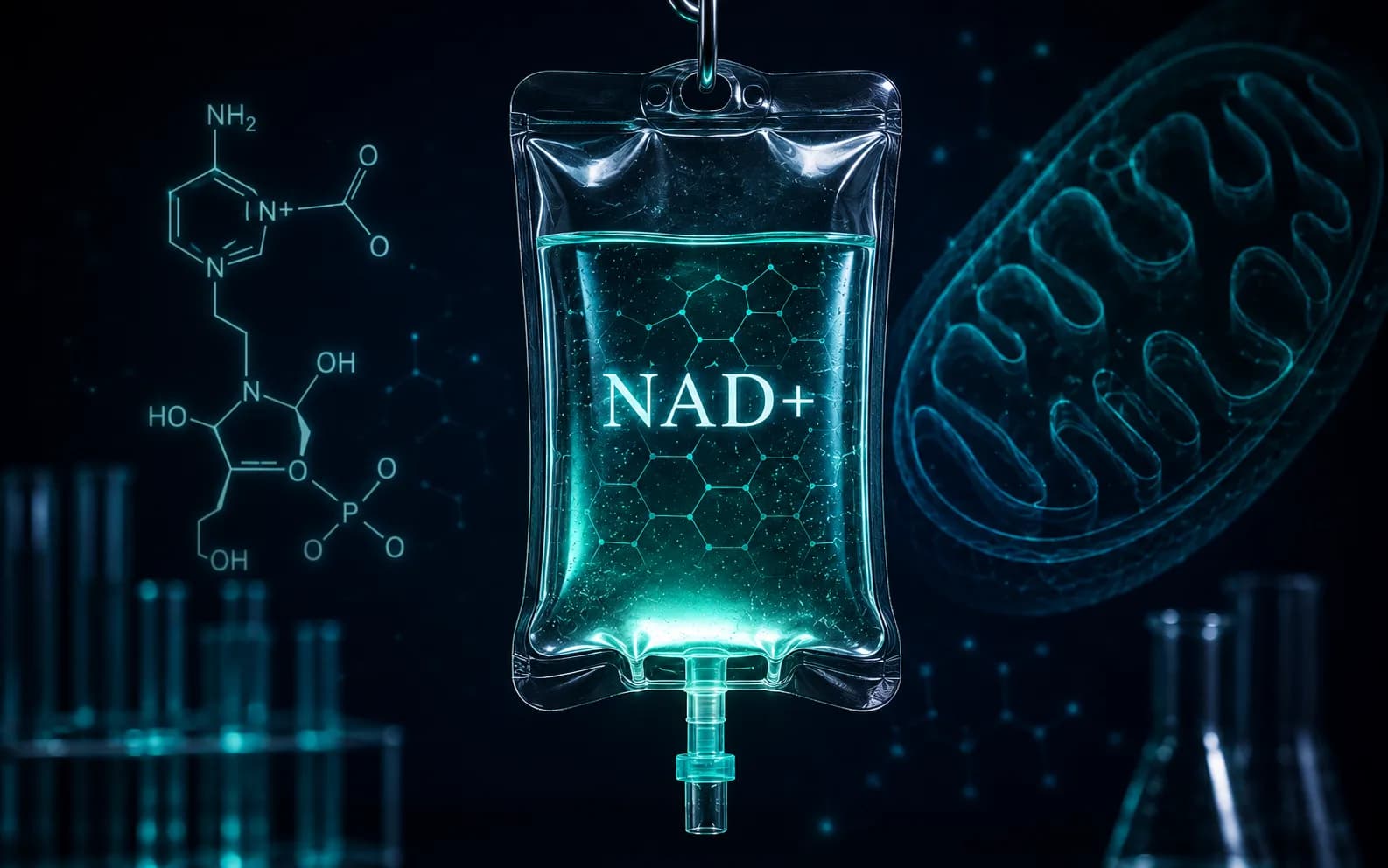
NAD+ is a coenzyme, not a peptide — set the category right first
Nicotinamide adenine dinucleotide — NAD+ — is a dinucleotide. Two ribose-linked nucleotides (one based on nicotinamide, one on adenine) joined by a phosphate bridge. It is not a chain of amino acids, has no peptide bond, and is not produced by solid-phase peptide synthesis. Listing it under a 'peptides' menu is a habit of online retail, not a reflection of its chemistry.[1][3]
Why the precision matters: peptide-quality language (HPLC purity, identity by LC-MS, sequence verification) does not map cleanly onto a small-molecule coenzyme. NAD+ quality is read through pharmacopeial purity tests, identity by NMR or mass spectrometry, and endotoxin or sterility tests for any injectable form — different methods, same standard of caring whether the molecule on the label is the molecule in the vial.[11]
This guide treats NAD+ as what it is: a coenzyme central to the deacetylation and DNA-repair work that defines current longevity biology, packaged in delivery formats that range from oral precursors to direct injection, with very different regulatory and pharmacokinetic profiles.[1]