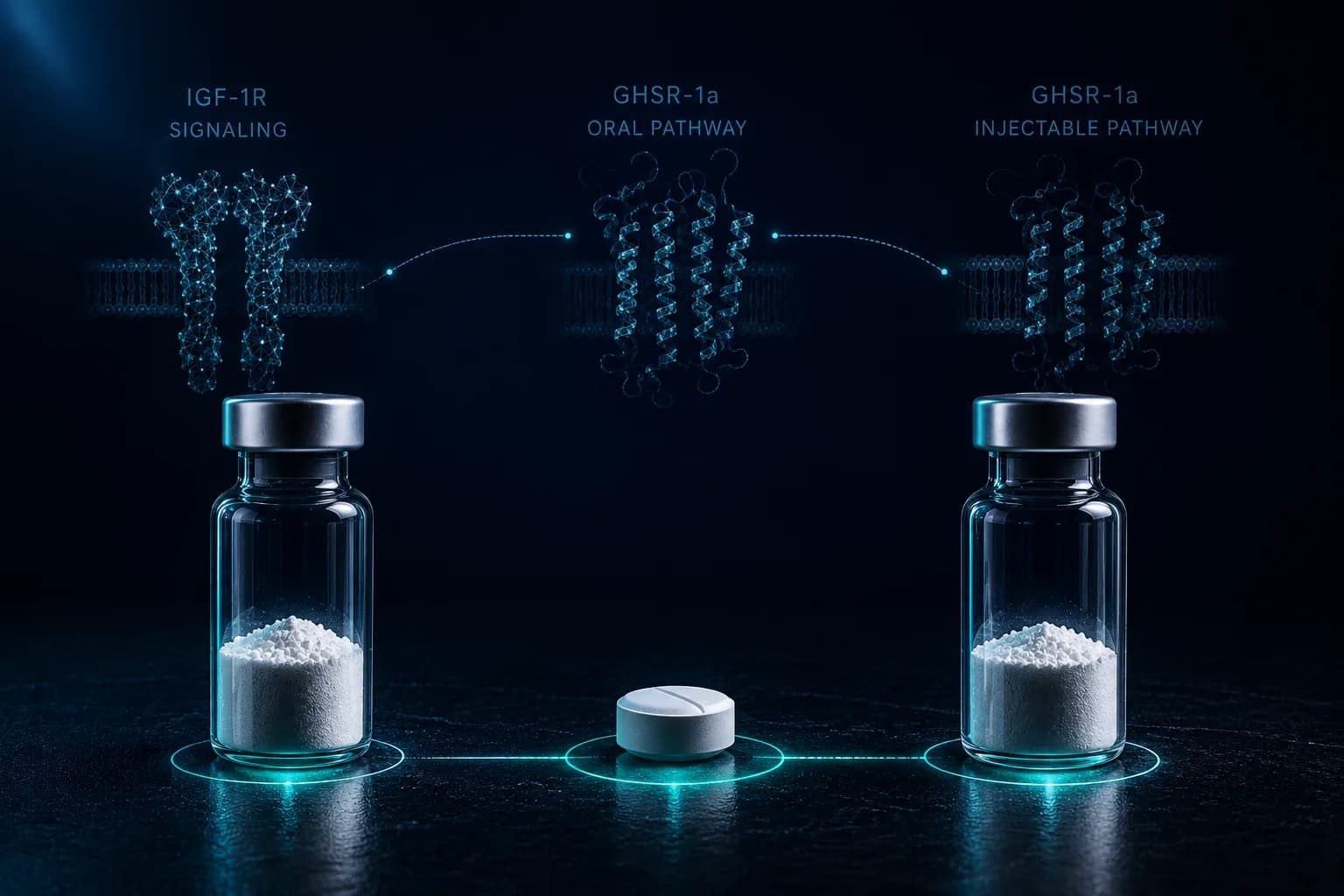
Three mechanisms that converge on the same axis
The growth-hormone / insulin-like growth factor-1 (GH/IGF-1) axis can be touched at three distinct levels. The hypothalamus releases GHRH (and ghrelin) that stimulate the anterior pituitary. The pituitary releases GH that travels to peripheral tissues. The liver, in response to GH, secretes IGF-1 that acts on IGF-1 receptors across muscle, bone, and connective tissue. Each level offers a different intervention point — and IGF-1 LR3, MK-677, and ipamorelin each enter at a different level.[9][1]
IGF-1 LR3 acts at the lowest level: directly on peripheral IGF-1 receptors as a synthetic recombinant ligand. It bypasses the pituitary entirely. MK-677 acts at the highest level above the pituitary: as an oral ghrelin-mimetic small molecule that stimulates GHSR-1a on the pituitary somatotrophs to release endogenous GH, which then drives endogenous IGF-1. Ipamorelin acts at the same level as MK-677 — GHSR-1a agonism — but as an injectable selective pentapeptide rather than an oral small molecule, with a far shorter half-life and a cleaner selectivity profile.[5][9][8]
That structural difference is the whole comparison. Saying 'all three raise IGF-1' is technically true and analytically empty — the route, time-action, side-effect profile, and clinical-evidence depth differ in ways that matter for research design.[4][1][7]